PRODUCT INTRODUCTION
We are the expert of endotoxin testing and endotoxin removal. We offer the complete solutions covering the whole range of endotoxin control, endotoxin monitoring and testing. Our products include laboratory research uses endotoxin removal kits and could be scaled up to industrial production scale columns. Lipopolysaccharide (LPS) is a bacterial endotoxin and a major constituent of the cell walls of gram-negative bacteria. Recombinant protein from E.coli usually contains high level of endotoxins. Removal of these endotoxins is highly necessary for downstream processes.
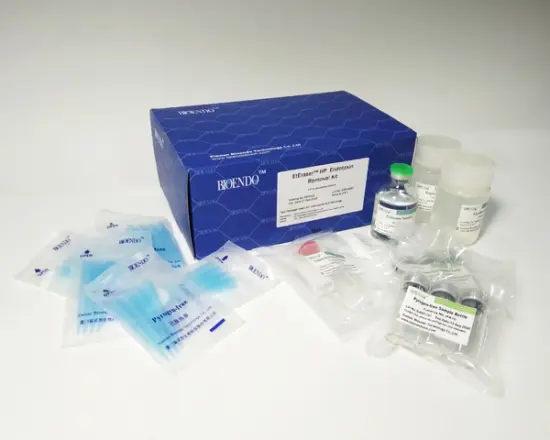
EtEraser HP Endotoxin Removal Kit are designed to remove endotoxin contamination from aqueous solutions. The kits contain endotoxin removal resin binds to reduce endotoxin levels in protein samples by ≥ 99% in less than 2 hours.
This endotoxin removal kit can be used in protein, DNA/RNA, polysaccharide and other biological samples.
Specifications
◆ high stability — does not affect the activity of most of the biological sample
◆ high protein recovery — >95% protein recovery for protein samples
◆ high removal efficiency — remove > 99% endotoxin, endotoxin level in the sample can be less than 0.1 EU/ml after endotoxin removing process
◆ wide application range — can beused in endotoxin removal for proteins, peptides, antibodies, vaccines, polysaccharide and other biological samples
EtEraser HP High Efficient endotoxin removal kit utilizes immobilized affinity ligand modified PMB to bind and remove endotoxin from aqueous solution. The modified PMB (polymyxin B) ligand is a high specific endotoxin binding ligand. The column has very high protein recovery rate for more than 95%. Endotoxin level in the sample can be less than 0.1 EU/ml after endotoxin removing process.
The kit includes a pre-packed endotoxin removal column 1.5 ml, Equilibration Buffer, Regeneration Buffer and pyrogen-free collection tube and tips. The column has a high binding capacity of > 2,000,000 EU / ml. This product may be reused up to five times if properly regenerated. The affinity resin is available inslurry and could be up-scaling in biopharmaceutical process.
Application
Et Eraser HP Endotoxin Removal Kit are designed to remove endotoxin contamination from aqueous solutions. The kits contain endotoxin removal resinbinds to reduce endotoxin levels in protein samples by ≥ 99 % in less than 2 hours.
This endotoxin removal kit canbe used in protein, DNA/RNA, polysaccharide and other biological samples.
Order information
| Catalog Number | Description | Kit Contents |
| ER0015 | EtEraserTM HP endotoxin removal kit |
EtEraserTM HP endotoxin removal pre-packed column 1.5 ml Equilibraion buffer Regeneration buffer Pyrogen - free colection tube Pyrogen - free tips |
| ER0100 | EtEraserTM HP endotoxin removal resin |
EtEraserTM HP endotoxin removal resin 10 ml Regeneration buffer Equilibration buffer
|