PRODUCT INTRODUCTION
Specifications
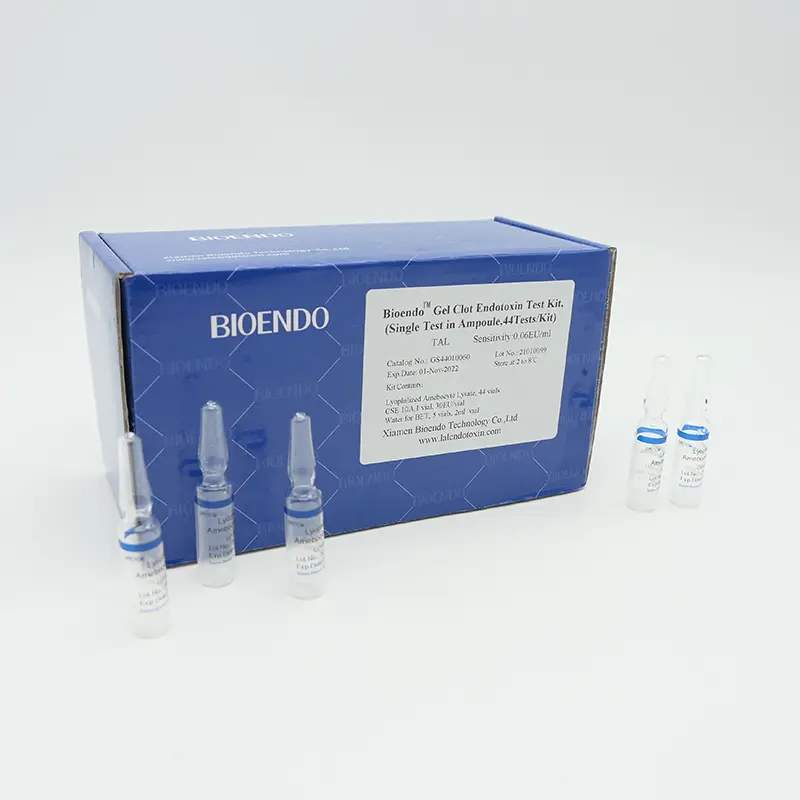
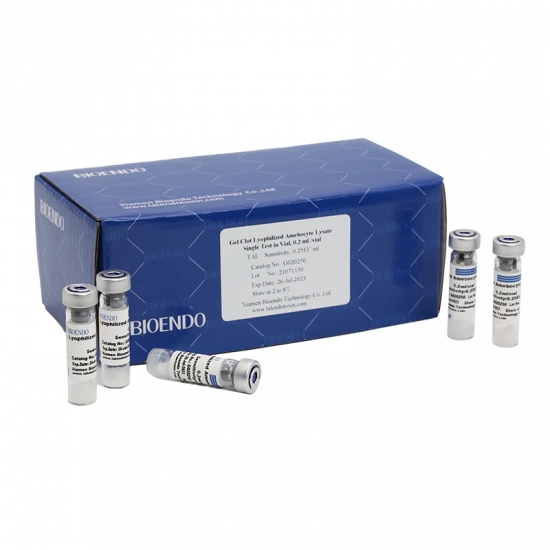
The Gel Clot Lyophilized Amebocyte Lysate (LAL) Single Test in Ampoule is a type of endotoxin test assay that is widely used in the pharmaceutical and medical device industries to detect the presence of endotoxins in various samples.
Here are some of the features of this single test endotoxin test assay:
1. Sensitivity: The Gel Clot LAL Single Test in Ampoule is highly sensitive and can detect endotoxin levels as low as 0.03 EU/mL.
2. Specificity: The test is highly specific to endotoxins and does not cross-react with other substances in the sample.
3. Convenience: The single test format of the Gel Clot LAL Single Test in Ampoule makes it convenient and easy to use, as it eliminates the need for preparing reagents and standard curves.
4. Stability: The lyophilized format of the test reagents provides excellent stability, allowing the test to be stored at room temperature for extended periods without compromising its performance.
5. Cost-effective: The single test format of the Gel Clot LAL Single Test in Ampoule is more cost-effective than other types of endotoxin test assays, such as the kinetic chromogenic LAL assay.
The Gel Clot LAL Single Test in Ampoule is a reliable, sensitive, and convenient method for detecting endotoxins in a wide range of samples, making it an essential tool for ensuring the safety and quality of pharmaceutical and medical device products.
Application
Single step endotoxin detection,
Not requires sophisicated microplate reader, normal endotoxin assay incubator or water bath is able to use.
Suitable for end-product endotoxin testing before product released.
Product sensitivity standardized according to US Pharmacopoeia criterion and China Pharmacopoeia criterion.
Order information
| Catalog No. | Sensitivity EU/ml | Description | Kit Contents |
| GS44010030 | 0.03 | Bioendo Gel Clot Endotoxin Test Kit, (Single Test in Ampoule, 44Tests/Kit) |
44 Gel Clot Lyophilized Amebocyte Lysate; 1 CSE 10A; 5 Water for BET , 2ml/vial |
| GS44010060 | 0.06 | Bioendo Gel Clot Endotoxin Test Kit, (Single Test in Ampoule, 44Tests/Kit) |
44 Gel Clot Lyophilized Amebocyte Lysate; 1 CSE 10A; 5 Water for BET , 2ml/vial |
| GS44010125 | 0.125 | Bioendo Gel Clot Endotoxin Test Kit, (Single Test in Ampoule, 44Tests/Kit) |
44 Gel Clot Lyophilized Amebocyte Lysate; 1 CSE 10A; 5 Water for BET , 2ml/vial |
| GS44010250 | 0.25 | Bioendo Gel Clot Endotoxin Test Kit, (Single Test in Ampoule, 44Tests/Kit) |
44 Gel Clot Lyophilized Amebocyte Lysate; 1 CSE 10A; 5 Water for BET , 2ml/vial |
| GS44010500 | 0.5 | Bioendo Gel Clot Endotoxin Test Kit, (Single Test in Ampoule, 44Tests/Kit) |
44 Gel Clot Lyophilized Amebocyte Lysate; 1 CSE 10A; 5 Water for BET , 2ml/vial |